Additive manufacturing (aka 3D printing) has a singular, strategic advantage in the medical device market: personalization.
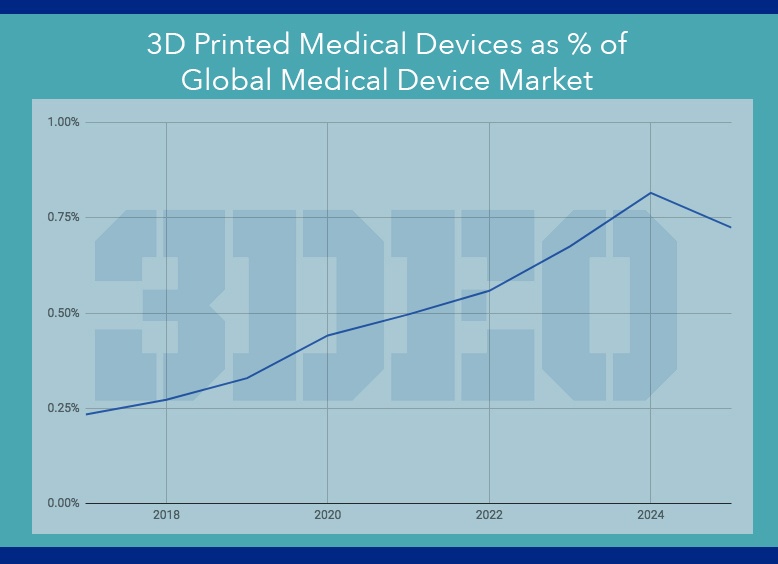
The global market for 3D-printed medical devices is expected to explode to nearly $10 billion by 2025, which represents an eleven-fold increase from the estimated $840 million market in 2017.
Even so, additive manufacturing (AM) will account for less than one percent of the global medical device market for the foreseeable future. But AM’s annual growth rate will outpace traditional manufacturing methods, thanks to personalized medical devices.
Medical Device Personalization: 3D Printing’s Special Sauce
Unlike traditional mass-volume production techniques, AM allows manufacturers to create complex geometries from unique digital designs.
Quite literally, AM technologies produce medical devices like prosthetics and other implantable devices that are sized to a patient’s specific dimensions and with materials and finished surfaces to promote biointegration.
Certain applications demand personalized devices. Consider the case of Penelope Heller, who needed a new sternum and a partial rib cage replacement because of a rare bone cancer (chondrosarcoma). She is the first patient in the U.S. to receive a 3D-printed sternum implant personalized to the specific geometric needs of her own skeletal system.
Speaking of bone cancer, it’s research into bone cancer treatment that’s spurring the development of just-in-time medical implants and surgical processes.
Researchers are fusing 3D printing technology and robotic surgery to manufacture and implant tailored bone replacements for patients, which requires a high level of personalization so the implants are an exact dimensional match and readily accepted by the patient’s skeletal system. The just-in-time process makes it possible for a surgeon to assess, model, and have the device produced and implanted during the same surgery.
Looking for a very public display of personalized medical devices in action? How about Game 4 of Major League Baseball’s World Series?
Five-year-old Hailey Dawson threw out the first pitch using her 3D-printed prosthetic hand, manufactured from 37 individual CAD files. Not only was the design personalized to her body dimensions, she also sported a customized version in Orioles Orange.
#throwlikeagirl
CHECK OUT TOP 4 IMPLANTABLE MEDICAL DEVICES
FDA Approval: Trying to Keep Pace with Innovation
Medical device innovation sometimes hits the brick wall of approval.
The Food and Drug Administration (FDA) oversees the approval of medical devices, and they are flooded with new devices daily. As the FDA struggles to manage the approval request process, they have developed allowances to expedite patient-based applications.
In other words, the FDA may allow an unapproved device if a patient can directly benefit from its use, and if that patient may suffer or die without it.
The program, known as Compassionate Use, is an example of need driving the approval process. In fact, in the example above of Penelope Heller and her sternum replacement implant, her medical team needed to proceed under the Compassionate Use program before they could perform the surgery.
Under the Investigational Device Exemption (IDE), the FDA specifically allows the use of an investigational medical device (even without clinical study) when a patient has a serious or life-threatening condition that can’t be remedied with approved treatments.
This steam-valve allowance from the FDA will allow innovation to continue at it projected blitzkrieg pace so that more patients can experience the life-saving benefits of 3D-printed medical devices.
3DEO for Medical Device Manufacturers
Using our Intelligent Layering™ metal additive process, 3DEO helps medical device manufacturers produce 3D-printed components cost-effectively – even in small volume batches. Streamlined design, set-up, and production helps you get your device to market sooner.
Contact us to learn more.